Overview of PVT Experiments
Below is a short summary of the most common PVT experiments with a short description of when they are used and what they information give. An important note is that the original intent of the PVT experiments was to try and emulate depletion of different reesrovir types. However, today PVT experiments are typically only used to develop fluid models.
Depletion Experiments
The depletion experiments described below are defined by their attempt to predict the reservoir behavior during the life (depletion and/or EOR) of the well / field.
Differential Liberation Expansion
Note
Main article is: Differential Liberation Expansion Experiment
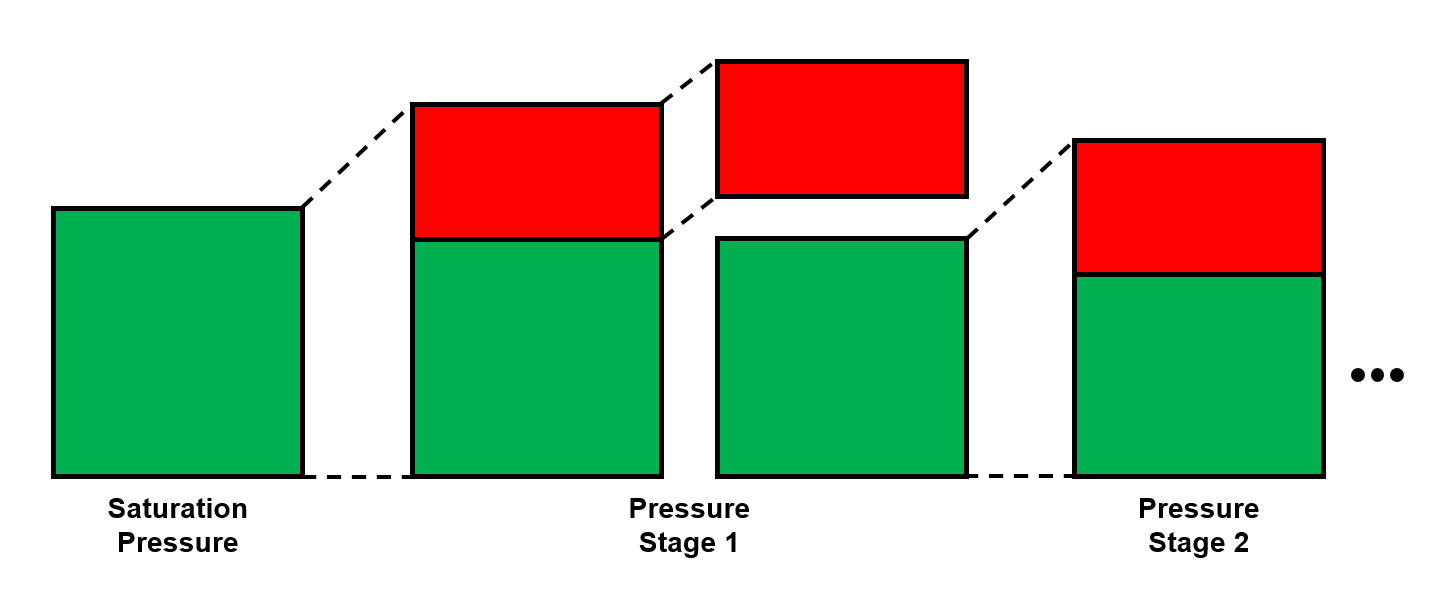
Figure 1: Schematic of a DLE experiment.
The differential liberation expansion (DLE) test is typically only used for black-oil fluids. The SPE Monograph Phase Behavior states that the DLE test is designed to emulate production of an oil reservoir and in turn, the results of the DLE test reflect the production properties. Specifically, the solution GOR and oil FVF are given as a function of pressure. This gives an indication of the composition variation in the life of an oil field.
The procedure for the DLE is given in Figure 1.
Constant Volume Depletion
Note
Main article is: Constant Volume Depletion Experiment
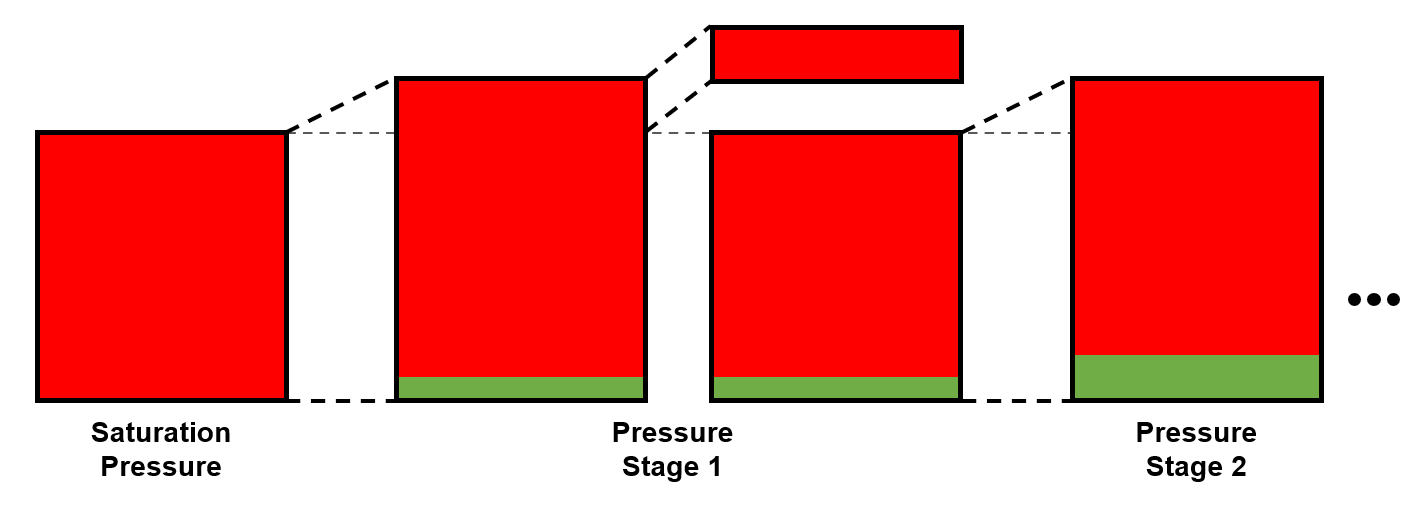
Figure 2: Schematic of a CVD experiment.
The constant volume depletion (CVD) test is typically only used for gas condensate fluids and volatile oils.
The CVD test provides several properties, among others, the liquid dropout curve and material balance data which can be used directly by reservoir engineers. The procedure for the CVD is given in Figure 2.
Constant Composition Expansion
Note
Main article is: Constant Composition Expansion Experiment
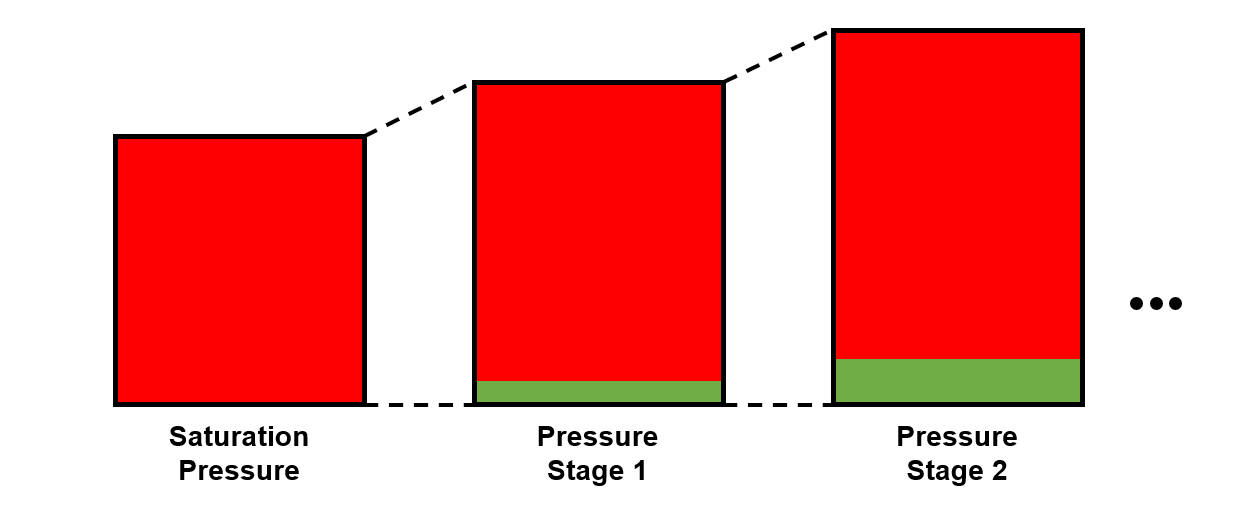
Figure 3: Schematic of a CCE experiment for an undersaturated saturated gas.
A constant composition expansion (CCE) test is mainly used to determine (1) the saturation pressure, undersaturated densities, isothermal compressibility and two-phase volumetric behavior at pressures below the saturation pressure.
The procedure for the CCE is given in Figure 3.
Surface Experiments
Surface experiments are defined here as a process that simulates the surface process. The two examples given here are (1) the true surface data and (2) laboratory multi-stage separation data.
Field Multi-Stage Separator
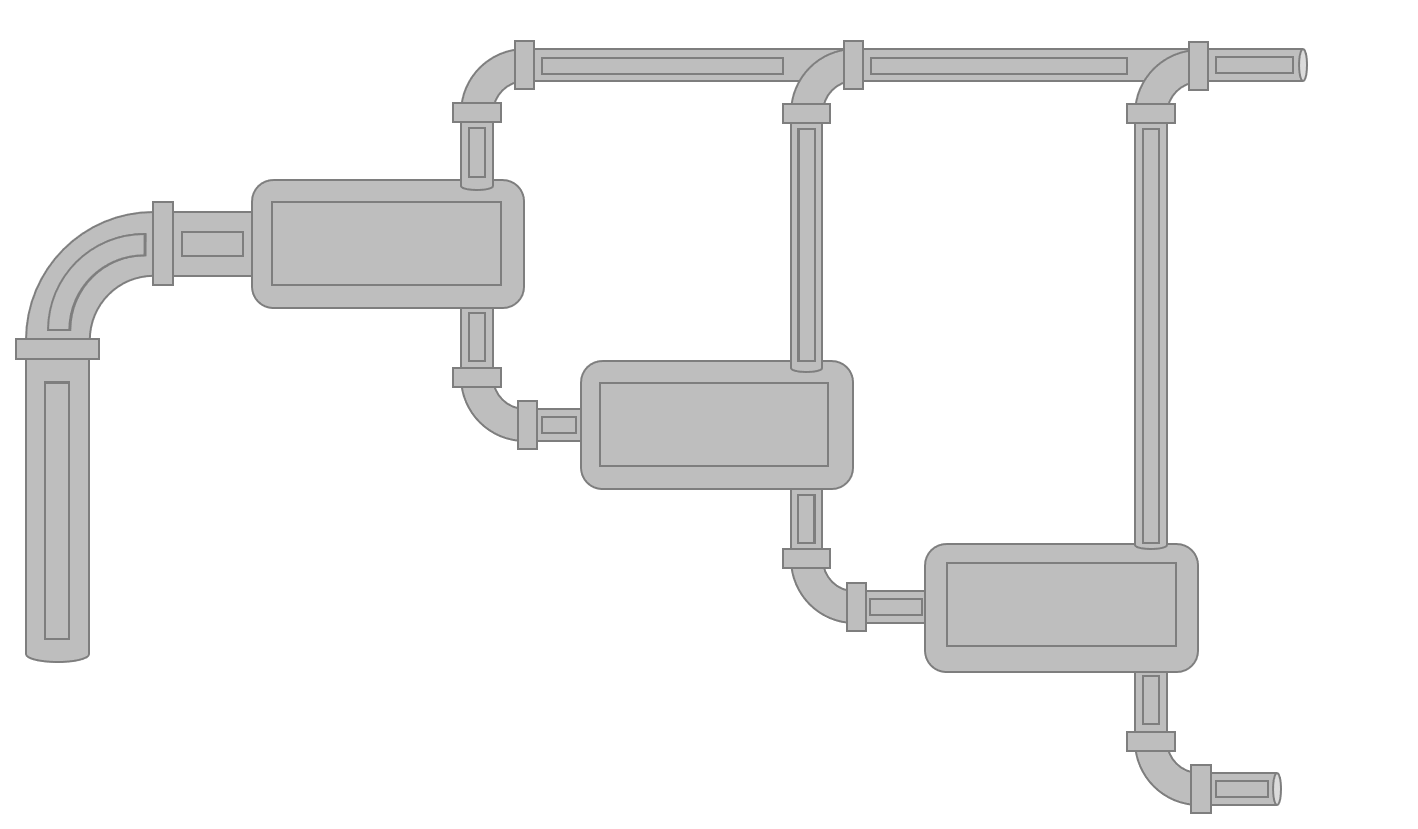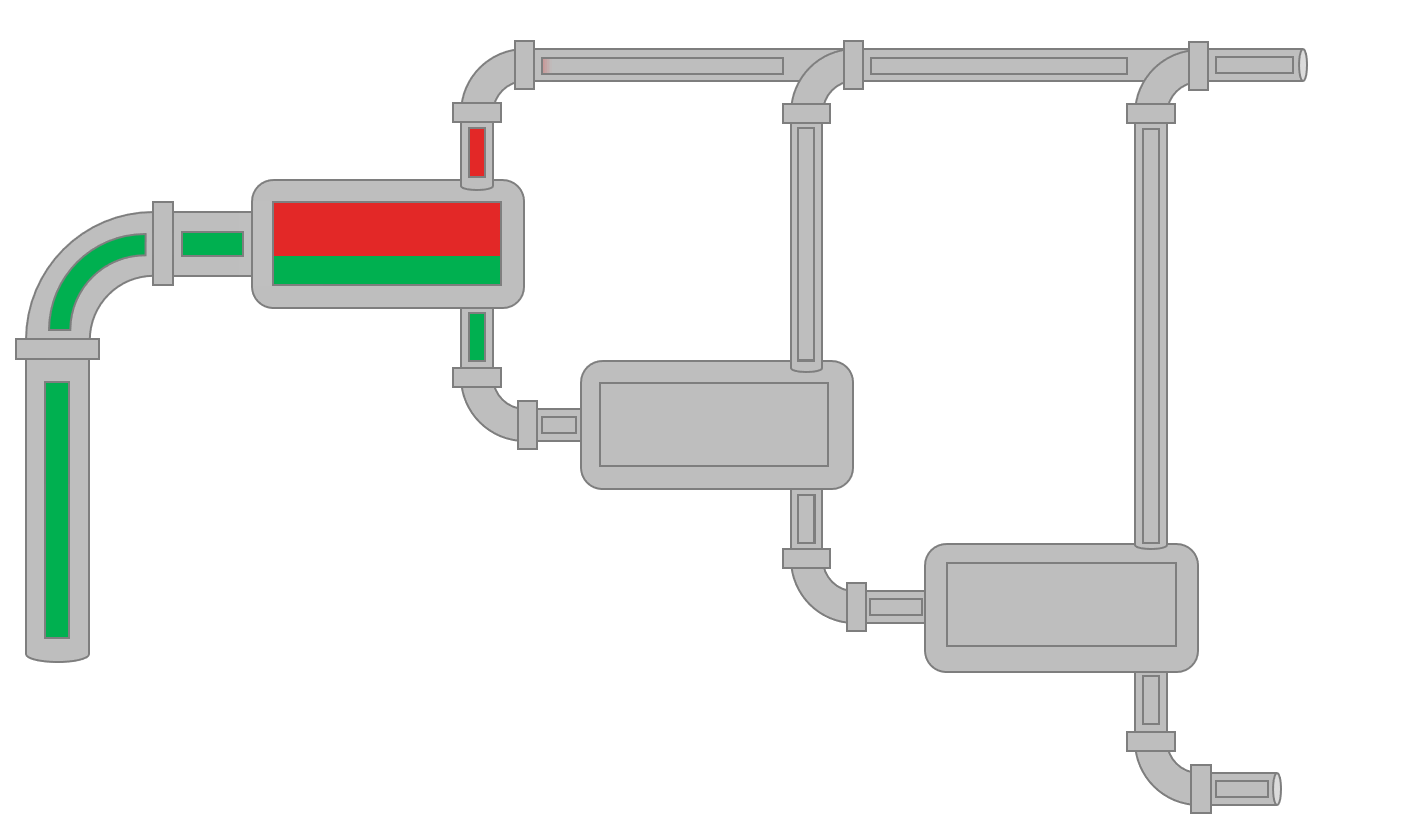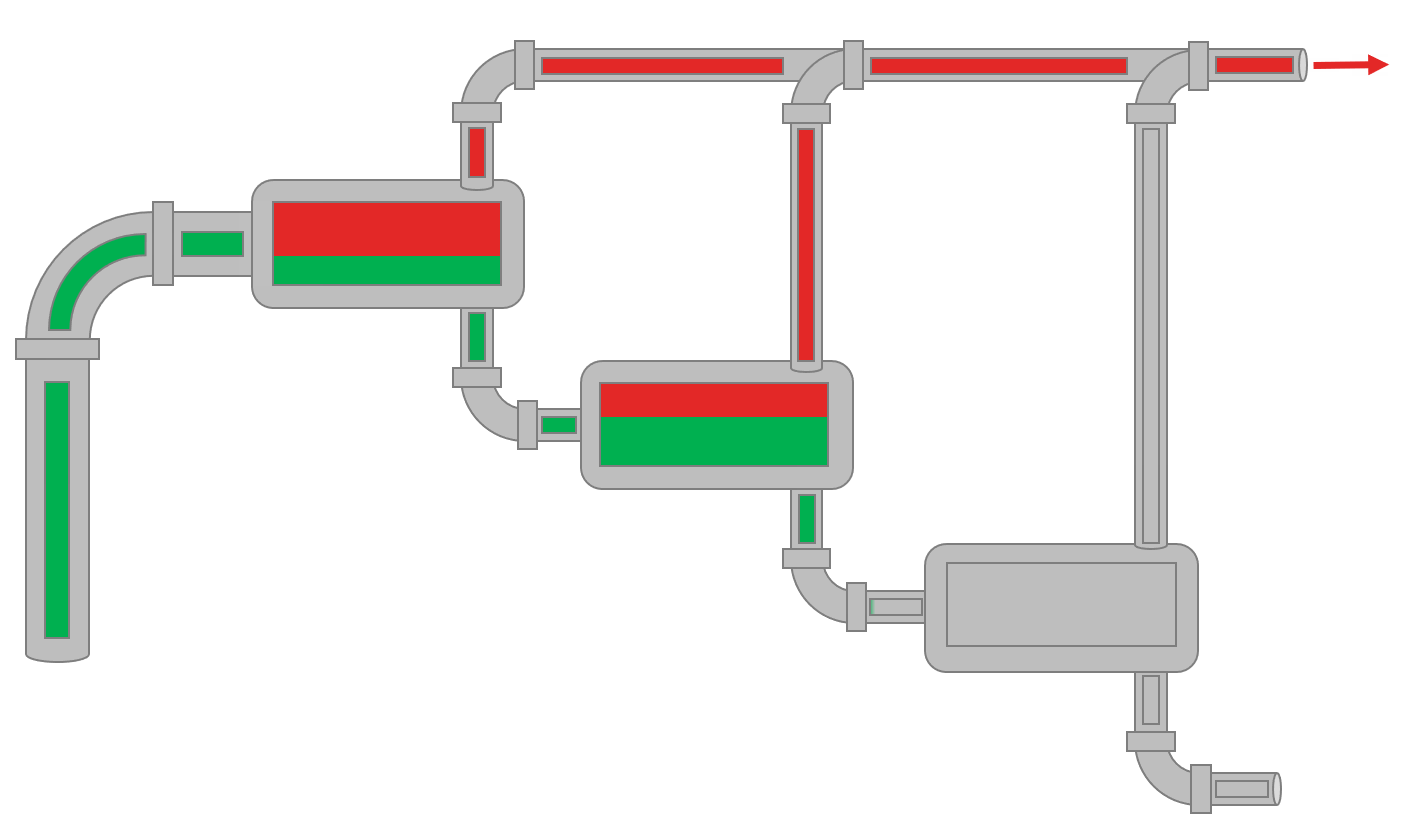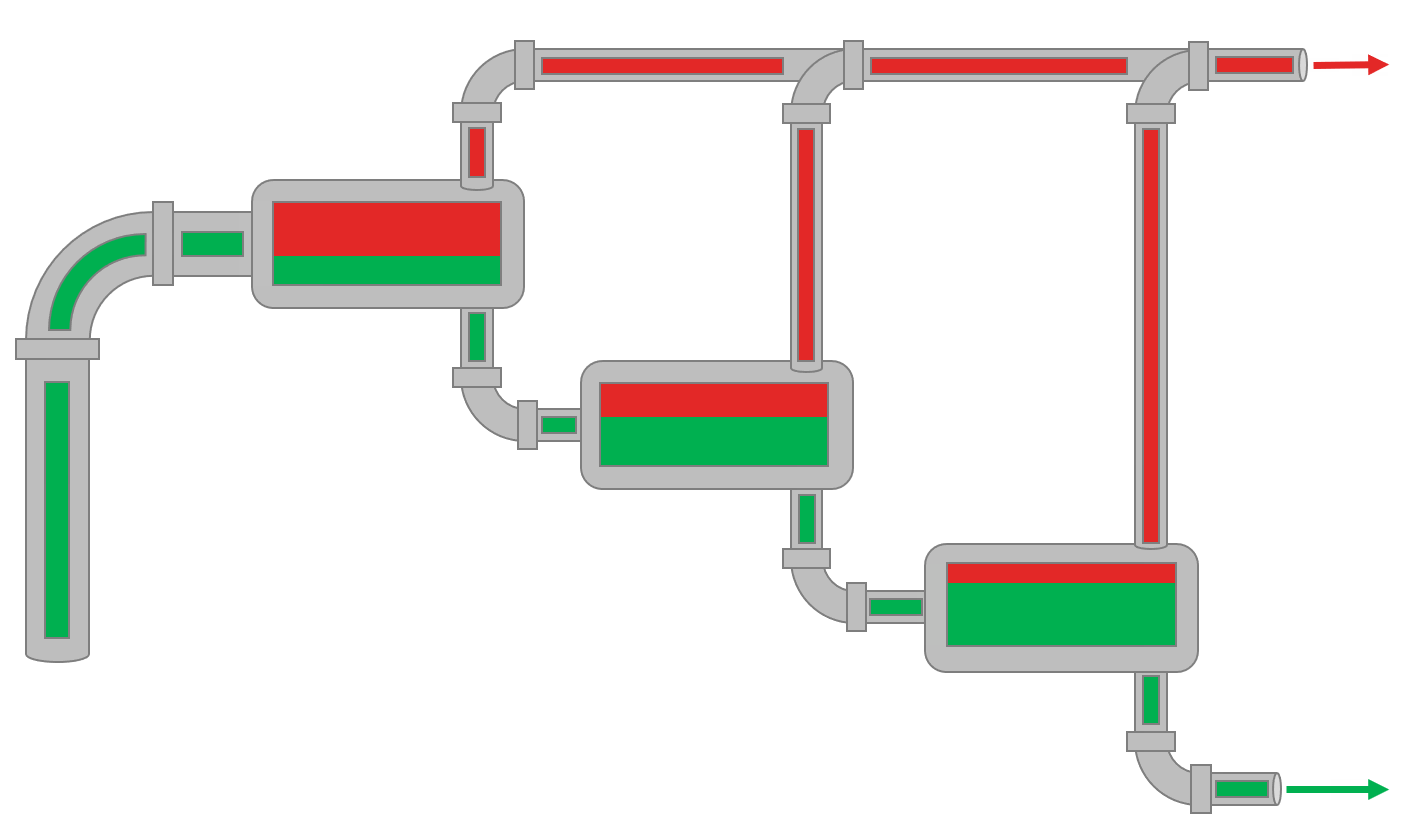
Animation 1: Animation of a field MSS procedure for a reservoir oil.
Note
The visual fluid volumes in Animation 1 are relative volumes.
The field multi-stage separator data (MSS) is simply a production test which are mandatory in many places to show the authorities what fluids are being produced in the various wells. Typically, separate production test separators are used when testing specific wells. The data given by separator production data is typically the surface compositions (up to \(C_{7+}\)) for gas (\(y_i\)) and oil (\(x_i\)), surface densities, GOR and \(C_{7+}\) properties like molecular weight (often back calculated from the average MW). Production test data varies a lot with respect to the country that the test is being taken based on government regulations and company standards.
The procedure for the field MSS test is given in Animation 1.
Laboratory Multi-Stage Separator
Note
Main article is: Laboratory Multi-Stage Separator Experiment

Figure 4: Schematic of a laboratory MSS experiment.
Laboratory multi-stage separator (MSS) tests are typically used to convert the DLE residual oil data to a stock-tank oil basis. It is also possible to use multiple laboratory MSS tests to optimize the surface process. The procedure for the laboratory MSS test is given in Figure 4.
Miscibility Experiments
The use of miscibility experiments is to determine how the reservoir fluid and an injection fluid will behave in a possible EOR project (displacement or mixing) and to what degree the mixtures will dominated by miscibility. The two example tests given below are indicate the two key types of miscibility, namely miscibility by first contact and multi-contact miscibility (also called developed miscibility).
There a whole range of different miscibility experiments and only a select few of them are summarized here.
Slim-Tube
Note
Main article is: Slim-Tube Experiment

Animation 2: Animation of slim-tube procedure with a condensing/vaporizing type miscibility.
The slim-tube experiment is a laboratory procedure which tries to predict and estimate developed miscibility of two fluids. In the procedure, an injection fluid is injected to a pipe filled with an initial in-place fluid.
Swell Test
Note
Main article is: Swell Test Experiment
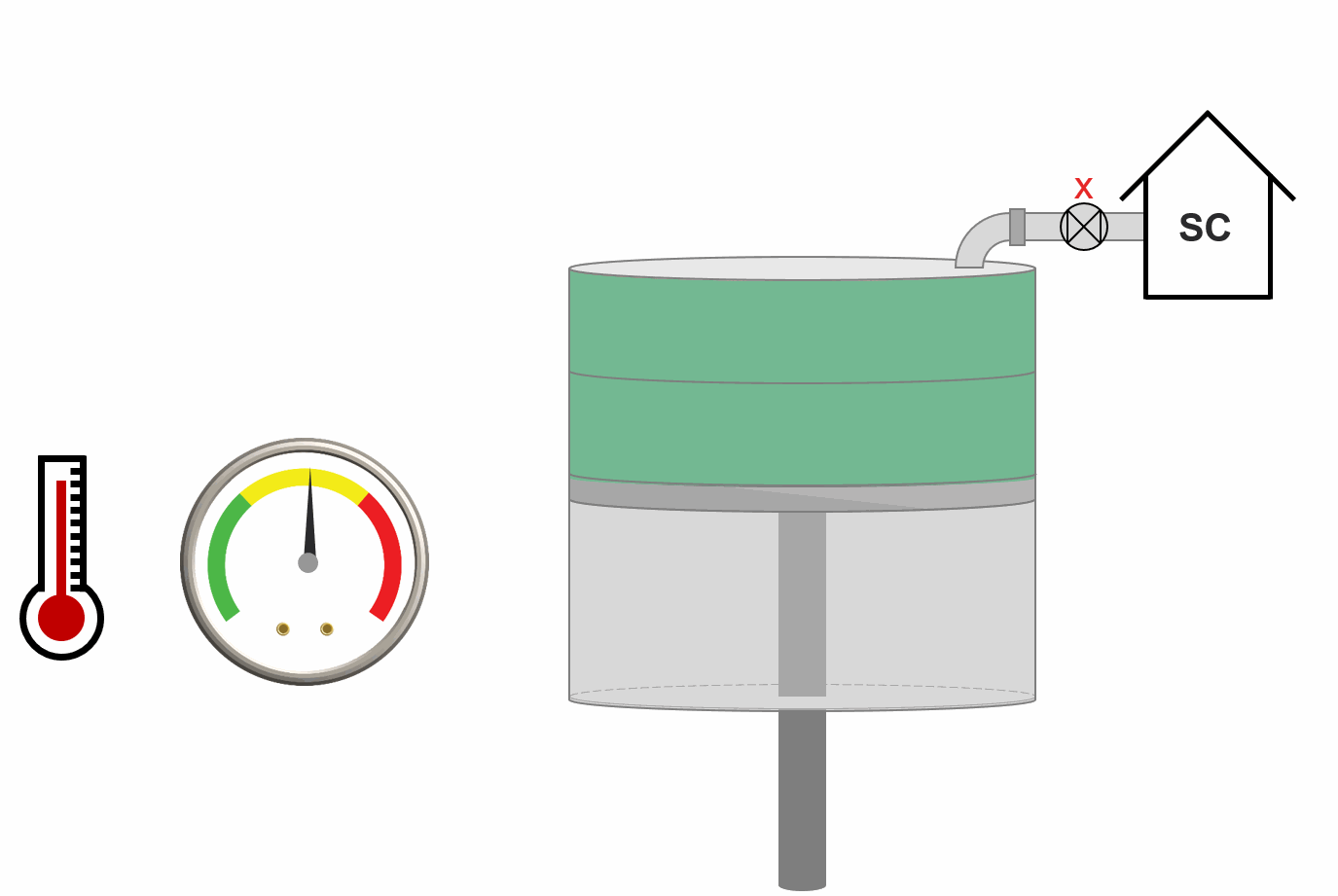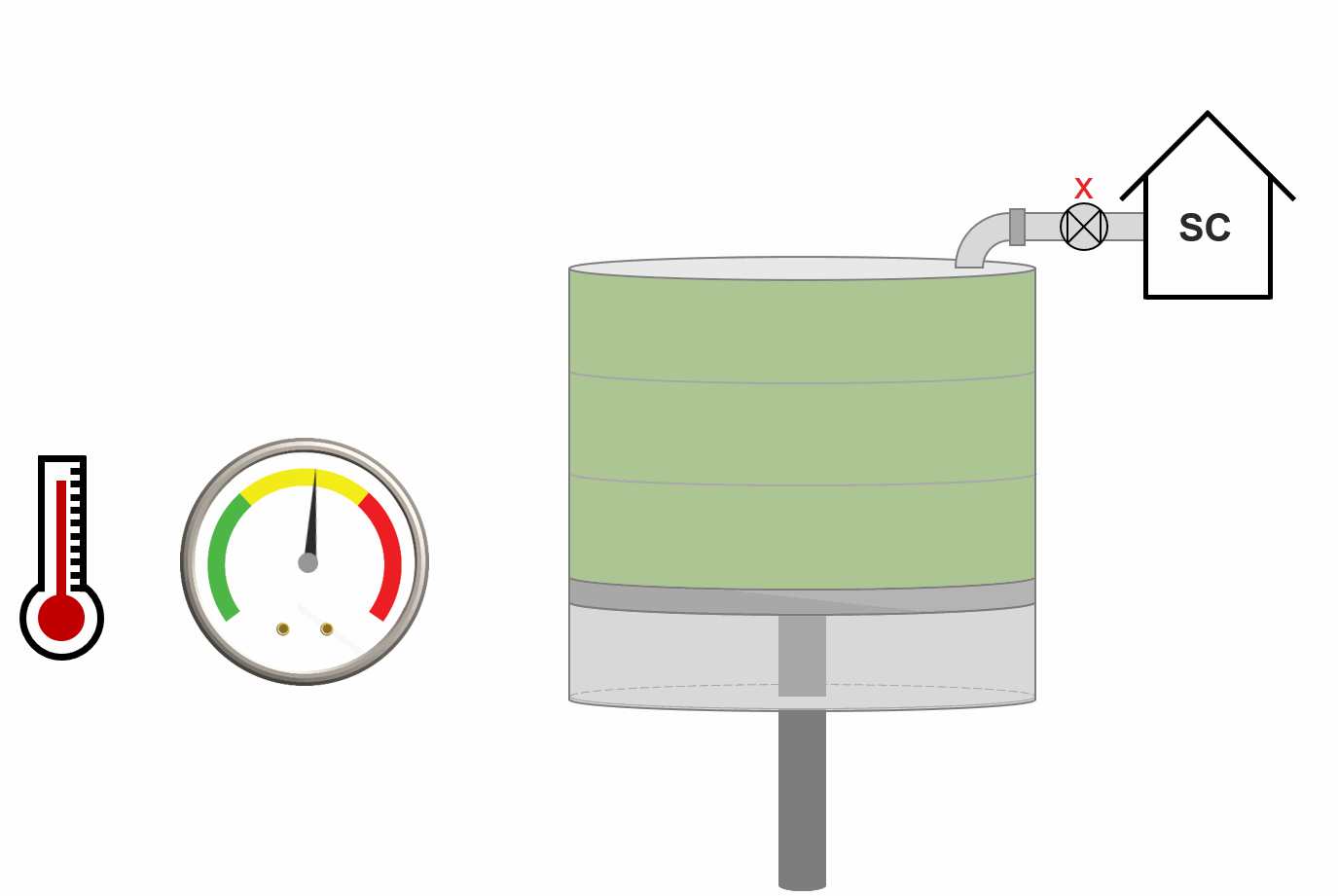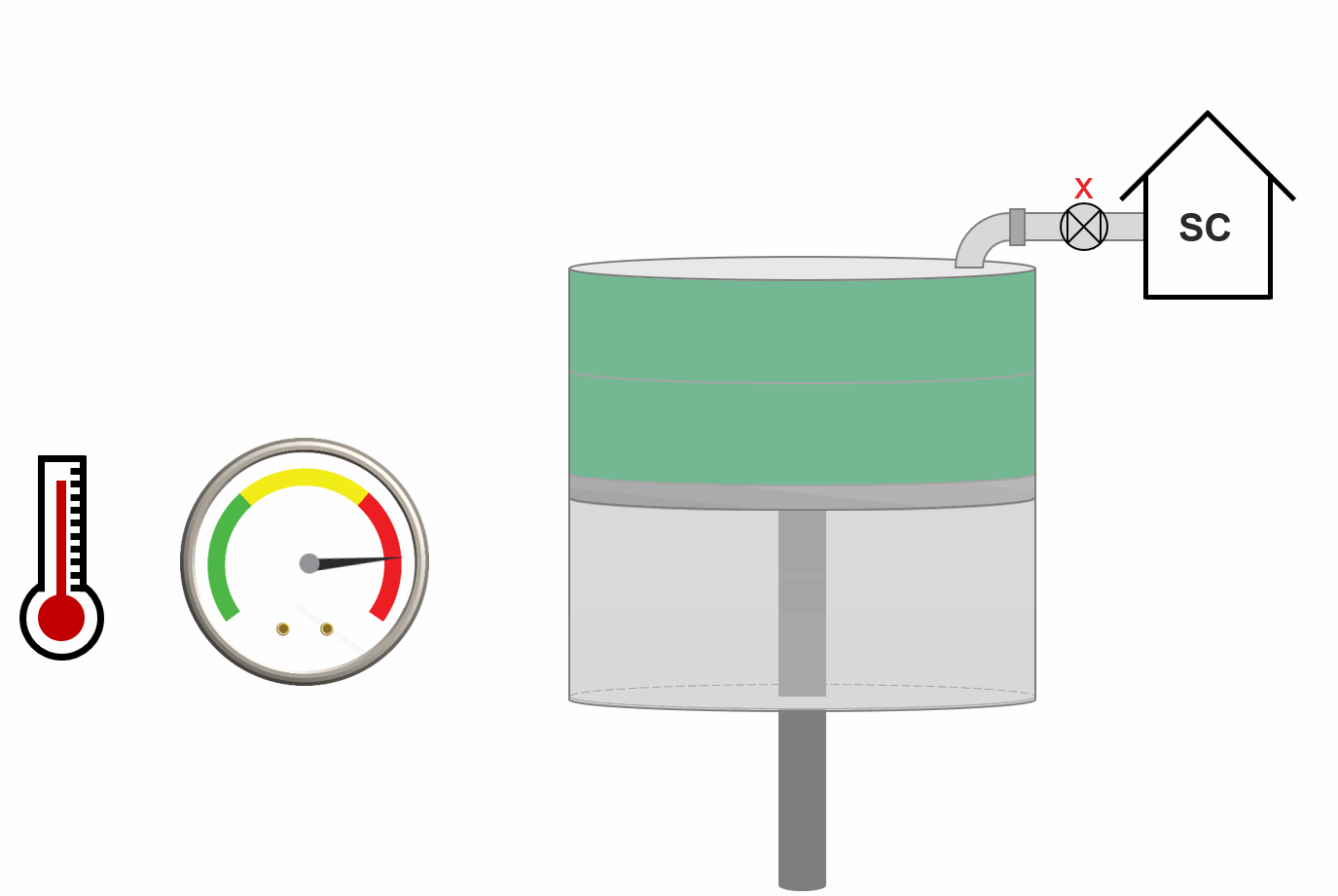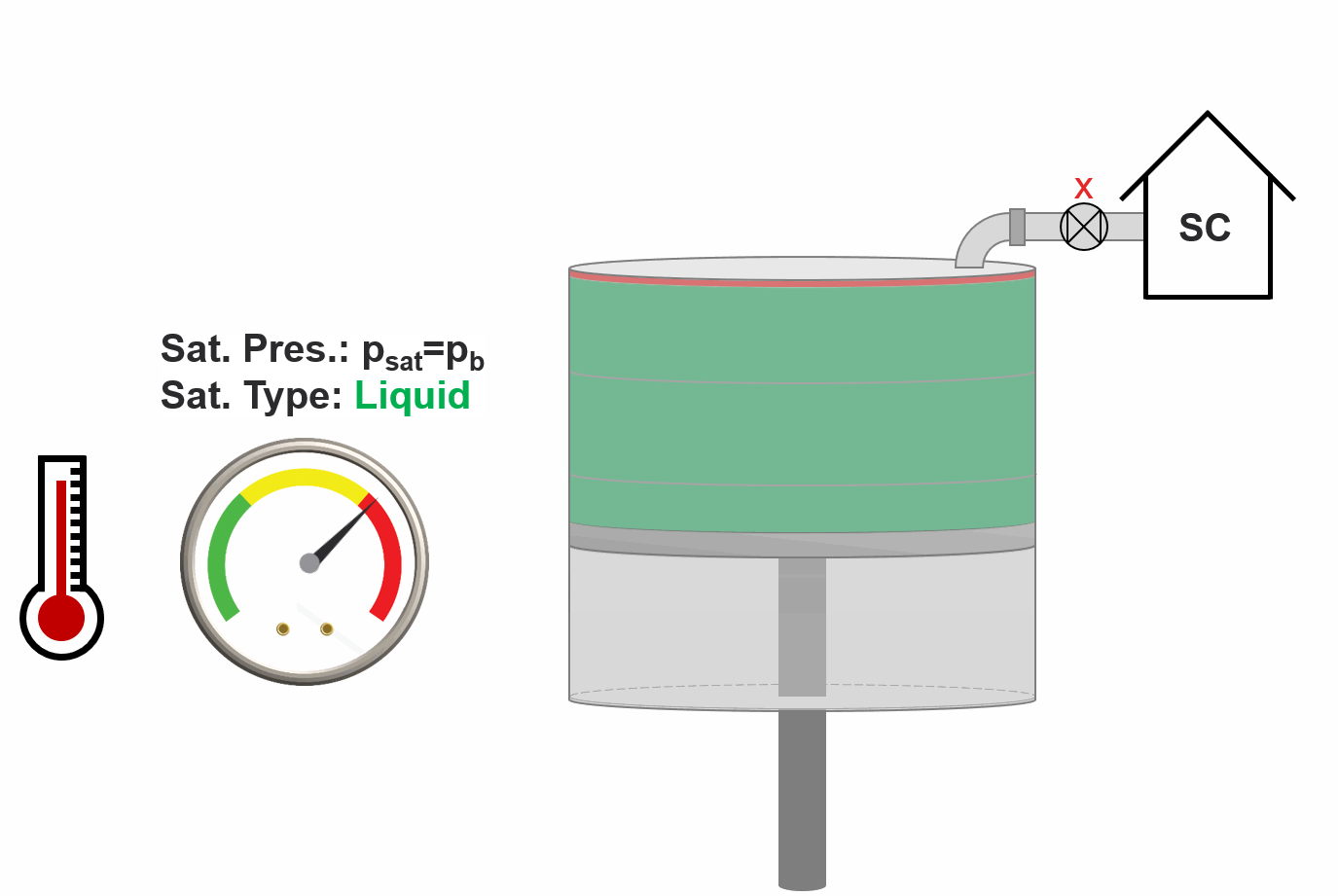
Animation 3: Animation of swell test procedure.
In the context of miscibility, a swell test can be used to estimate the first constact minimum miscibility pressure (MMP\(_{FC}\)).
The approach of the swell test is to take an initial volume of the reservoir fluid (typically a reservoir oil) and (1) add an incremental amount of injection gas (2) estimate the saturation pressure and saturation type (bubble-point or dew-point). This procedure is done for an increasing amount of injection gas and the saturation pressure and type is stored. Figure XX shows an example of a swell test result where the relative moles injected is just a measure of the amount of injection gas injected. The procedure is also animated in Animation 2.
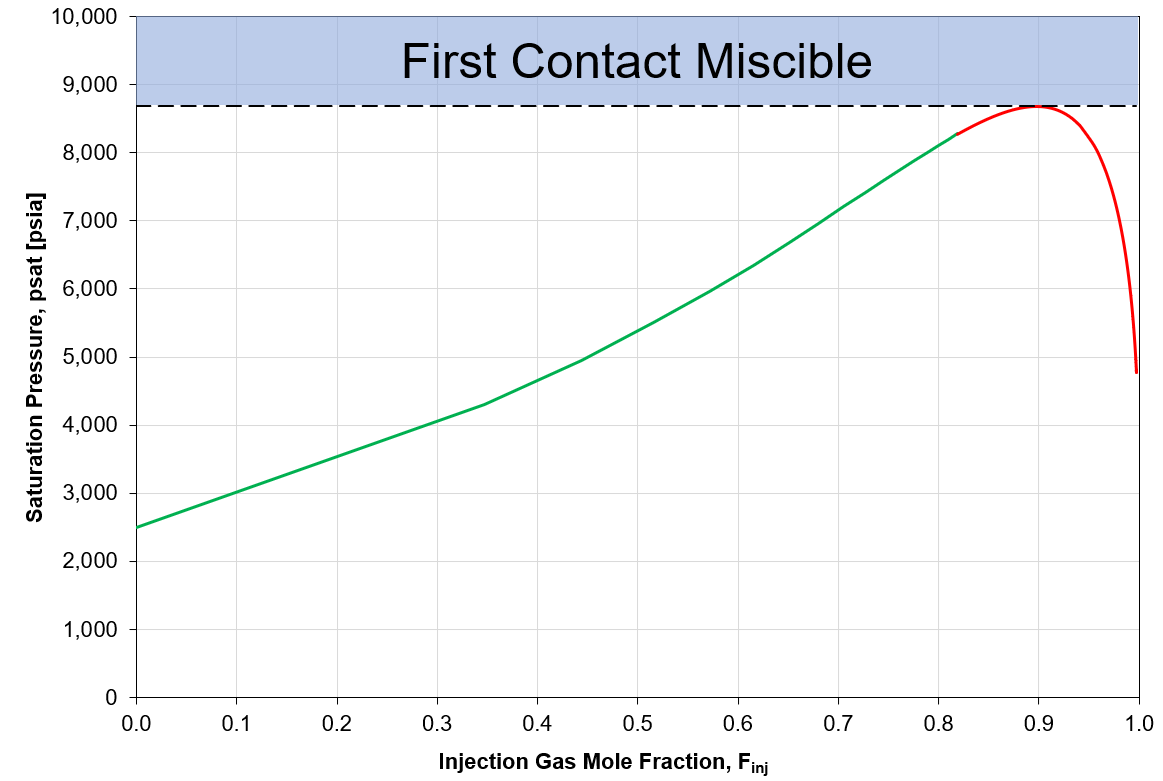
Figure 5: Example of swell test data and indicated MMP by first contact.
Once the swell test data is found, the MMP by first contact is determined by the largest value of the swell test data. Note that there is some experimental evidence showing asymptotic behavior of the swell test curve (i.e. the curve "blows up") which yields no MMP\(_{FC}\).